This article outlines the acute danger posed by all current SARS-CoV-2 vaccines as revealed by recent scientific research. This research is reviewed with citations and links to their sources. In addition, the research raises a number of serious long-term health implications that may only now be beginning to emerge. The reader is encouraged to review the source material on their own. While some is only in preprint form, much of it is peer-reviewed in respected journals.
How the SARS-CoV-2 Virus Works
A virus or “virion” is an assembly of proteins encapsulating the genetic code necessary to make a copy of itself; however, it lacks the cellular machinery to make such copies. As part of its structure, it contains surface proteins that have evolved to bind to the surface receptors of a host’s cells. Once binding occurs, the virion’s genetic material, either RNA or DNA, is injected into the host cell where it hijacks the host’s cellular replication machinery to make copies of itself.
In the case of the SARS-CoV-2 virus, the virion is a globular structure encapsulating genetic material called messenger RNA (mRNA). On its surface are protruding structures called spike proteins, giving the appearance of a crown of thorns – hence corona virus. Figure 1 is an image of what the SARS-CoV-2 virion looks like. Included is a software rendering of how the (trimeric) spike protein is configured in terms of its structural components.

The SARS-CoV-2 virion has an exceptionally strong binding affinity for a human cellular receptor known as ACE2 (Read Samavati (2020)[37] to learn about its function). ACE2 is found on the surface of epithelial cells which form the protective surface layer for the major organs, systems and glands of the body, especially the lungs. As well, ACE2 occurs on endothelial cells that line the inner surface of blood vessels and the heart. These are the major areas where the COVID-19 pathology is expressed.
The ACE2 receptor acts as a security gateway allowing essential chemicals and enzymes to enter the cell. They only allow some very specific molecules that fit the receptor, much like a key in a lock. Figure 2 shows how the SARS-CoV-2 virus spike protein, frequently referred to as ‘S protein’ or sometimes just ‘S’, has adapted to fit the ACE2 receptor to gain entry into the cell.
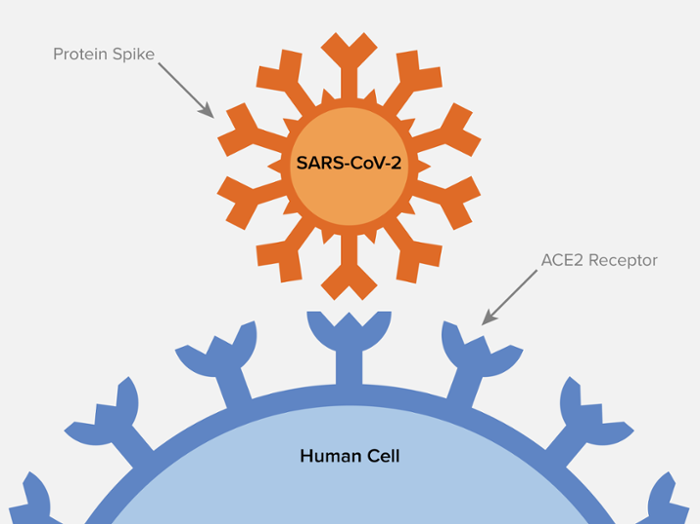
When the virion binds or fuses with the ACE2 receptor, it inserts its mRNA into the host cell. Replication of the virion ensues. The infected host cell eventually commits suicide or is killed by the host’s immune system, releasing the viral copies into the host’s body to infect other cells.
The key point is that the virus is able to inject into a host cell, the genetic code from which the infected cell produces complete copies of the virus.
How the SARS-CoV-2 Vaccines Work
There are two types of vaccines in widespread use in the West, mRNA vaccines (Moderna and Pfizer) and adenovirus vaccines (Astrazeneca and J&J). They differ in the type of delivery system that they use but the end result is the same: the inducement of the body to manufacture SARS-CoV-2 spike proteins. While some disagree with the use of the term ‘vaccine’ preferring ‘gene therapy’ instead, this is not an issue that we wish to debate, using ‘vaccine’ for convenience.
All SARS-CoV-2 vaccines are experimental[15][16], initially authorized for emergency use only in the US and other countries. The reason they do not have full approval for general use is that no long-term studies, known as longitudinal studies, have been completed for any of them. Such studies usually take a minimum of two years and as much as 7 years (watch the videos in this article). They are designed to detect long-term adverse effects. It should not be a surprise then as new adverse effects are reported in the media for these experimental vaccines.
All four vaccines take a segment of the SARS-CoV-2 genome that encodes the virion’s S protein. The S protein has two sub-units, S1 which contains the receptor binding domain (RBD) or part that is able to bind to or ‘plug into’ ACE2, and sub-unit S2. They then combine this mRNA strand with some mechanism or system that can inject the genetic code into a host cell for new copies of S to be made.
Astrazeneca and J&J use an innocuous virus of the adenovirus class for which the viral DNA has been removed and replaced with the SARS-CoV-2 S protein RNA segment after conversion to its DNA equivalent form. When this adenovirus infects a host cell, the cell manufactures copies of the S protein and not the adenovirus itself.
The Moderna and Pfizer vaccines use a different approach for cell ‘infection’. They attach the mRNA for the S protein to a lipid nanoparticle[20] which is able to penetrate a cell wall, introducing the mRNA that is attached to it. Replication of the S protein then ensues.
The key point is that all four vaccines infect a host cell in ways similar to the SARS-CoV-2 virus. The difference is that the infected cell produces copies of the S protein only and not a functional SARS-CoV-2 or adenovirus virion. It should be noted that the S protein contains no viral RNA and cannot be detected by the standard PCR test which has been designed to look for RNA fragments.
In the remainder of this article emphasis in quoted passages has been added by ourselves to draw attention to key point. Also, insertions in [brackets] are added to provide brief explanation of aspects of the quoted text for clarity and simplicity.
Two Critical Problems With These Vaccines
Recently, two properties of the vaccines have emerged as a cause for present and future concern. One is the mobility in the body of the vaccine delivery system after injection. The other is the pathogenic toxicity of the S protein that the vaccines are designed to produce. These properties are responsible for a number of adverse effects. To get an overview of the problem consider this 10-minute audio interview with Dr Byram Bridle.
Dr Bridle states that “the spike protein on its own is almost entirely responsible for damage to [the] cardiovascular system if it gets into circulation“. He also cites research that shows that the lipid nanoparticles as well as the S protein itself cross the blood-brain barrier (BBB), and the S protein accumulate in the spleen, bone marrow, liver, adrenal gland, and particularly heavily in the ovaries. We review the science supporting these and related claims in the remainder of this article.
The Spike Protein As a Pathogenic (Toxic) Protein
Suzukiand and Gychka (2021)[27] warned that the S protein may have long-term effects on the body. This requires complete longitudinal studies that we don’t have as well as ongoing monitoring by the medical community for emergent patterns of diseases implicated by S protein research. They note that:
it is important to be aware that the spike protein produced by the new COVID-19 vaccines may also affect the host cells. We should monitor the long-term consequences of these vaccines carefully, especially when they are administered to otherwise healthy individuals.
Suzukiand and Gychka (2021)[27]
In an early preprint, Avolio et al (2020)[29] show “for the first time, that the recombinant S protein alone elicits functional alterations in cardiac [pericytes] PCs“. Pericytes are cells connected to blood vessel endothelial cells (described above) and are essential for development of the vasculature or blood vessels of the central nervous system (CNS). The implications for the CNS are covered later under ‘Neurodegenerative Implications‘.
Khan et al (2021)[34] observed that the the S protein caused the release of chemicals called cytokines that cause inflammation in the affected tissue. When released in quantity, they cause what is called a cytokine storm, the cause of most of the lung damage from the SARS-CoV-2 virus. They note that the:
spike (S) protein potently induces inflammatory cytokines and chemokines including IL-6, IL-1ß, TNFa, CXCL1, CXCL2, and CCL2, but not IFNs in human and mouse macrophages.
Khan et al (2021)[34]
Keep in mind that it is the inflammatory response in the lungs along with clotting that results in the respiratory damage and this can be caused by the S protein alone without the presence of the virus.
Referring back to Dr Bridle’s interview, a seminal paper from the Salk Institute[22] identified the toxicity of the spike protein. As the authors state:
we show that S protein alone can damage vascular endothelial cells (ECs) by downregulating ACE2 and consequently inhibiting mitochondrial function.
Lei et al (2021)[22]
In other words, the S protein generated by vaccines damages the vascular walls of all elements of the circulatory system from heart muscle to micovasculature (very small) capillaries in the brain. By binding to the ACE2 receptors of endothelial cells lining the blood vessels as anticipated by Suzukiand and Gychka, it prevents them from performing proper essential functions[37] causing cellular damage leading to cell death. Blood clot causing stroke are also a possible result. As a Salk Institution news release states, the research:
shows conclusively that COVID-19 is a vascular disease, demonstrating exactly how the SARS-CoV-2 virus damages and attacks the vascular system on a cellular level.
Salk News (2021)[35]
Alexander et al (2021)[26] present information from several studies that indicates the S protein alone is responsible for vascular damage throughout the body and should therefore be considered a “toxic endothelial pathogen“.
Farsalinos et al (2020)[8] found that the S protein had genetic sequences similar to those found in the neurotoxins of snake venom, suggesting that the S protein should be studied for neurotoxic properties also. They
identified a “toxin-like” amino acid (aa) sequence in the Receptor Binding Domain [RBD] of the Spike Glycoprotein of SARS-CoV-2 (aa 375-390), which is homologous to a sequence of the Neurotoxin homolog NL1, one of the many snake venom toxins that are known to interact with nicotinic acetylcholine receptors (nAChRs).
Farsalinos K, Eliopoulos E, Leonidas DD, et a (2020)l[8]
In summary, above researchers have identified, as outlined in several studies, that the S protein generated by the vaccines is a pathogenic toxin. Some of its effects are immediate, but as we shall show, potential long-term consequences are of grave concern.
The Mobility of the Vaccine and the S Protein Throughout the Body
In an autopsy of a patient who died after a first dose vaccination but exhibited no morphological signs (meaning symptoms) of the disease SARS-CoV-2, Hansen et al (2021)[30] found:
Postmortem molecular mapping by real-time polymerase chain reaction [RT-PCR test] revealed relevant SARS-CoV-2 cycle threshold values in all organs examined (oropharynx, olfactory mucosa, trachea, lungs, heart, kidney and cerebrum) except for the liver and olfactory bulb.
Hansen et al (2021)[30]
While it cannot be ruled out that the patient may have had the COVID-19 disease, the fact that he had no symptoms and that there was no positive PCR response from the olfactory bulb (nose), a common portal of entry for the virus, supports the hypothesis that the mRNA found in almost all organs is from the vaccine itself. This would imply a high mobility of the vaccine after injection.
However, a report by Pfizer[1] to a Japanese regulatory agency was obtained by Dr. Bridle and colleagues which addressed the issue of the Pfizer BNT162b2 vaccine mobility or pharmacokinesis (PK). The report appears to be a machine translation and contains places where there is some discontinuity of text.
For the study, Pfizer attached an enzyme called luciferase to the lipid nanoparticle (LNP) of the vaccine. This addition to the BNT162b2 (vaccine) LNP causes it to glow when stimulated allowing for detection and quantitative measurement. This modified vaccine was injected into muscle tissue of mice and rats.
In some parts of the study, the LNP was tagged with a radionuclide (a radioactive marker) instead of luciferase allowing for quantitative radioactivity measurements. Key points of this study[1] are quoted below:
- No internal PK study [of BNT162b2] was performed.
- As a result, ALC-0315 and ALC-0159 [both ‘functional’ lipids encapsulating the mRNA] were shown to be rapidly distributed from the blood to the liver.
- From the above nonclinical pharmacokinetic evaluation, it was shown that LNP that reached the circulating blood is distributed in the liver. In addition, metabolism and fecal excretion may be involved in the disappearance of ALC-0315 and ALC-0159, respectively. It was suggested.
- In both males and females, the radioactivity concentration was highest at the administration site at all measurement points. The radioactivity concentration in [blood] plasma was the highest 1 to 4 hours after administration. Also, mainly the liver, spleen, adrenal glands and … the ovaries was observed, and the highest radioactivity concentration in these tissues was 8 to 48 after administration.
- The total radioactivity recovery rate for doses other than the administration site is the highest in the liver (up to 18%). Significantly lower in the spleen (1.0% or less), adrenal gland (0.11% or less) and ovary (0.095% or less) compared to the liver.
- the in vivo [animal study] expression distribution of the antigen [S protein] encoded by BNT162b2 is considered to depend on the LNP distribution
In discussion of this document, point 1 is apparently a statement that Pfizer did not consider human PK tests necessary. Points 2 and 3 indicate that a major distribution of the vaccine LNP happened via the circulatory system to the liver. Points 4 and 5 present an assay of the amount of LNP transferred to various organs in the body. In fact, table 2.6.5.5A and B show quantitatively, the distribution of LNP to virtually every organ in the bodies of test animals. The final point 6 equates the distribution of the LNP to the vaccine itself.
The experimental procedure was to innoculate a test group of enough specially bred mice or rats to create statistically significant results. The animals would be euthanized at a particular time point, disected, and their body parts analysed for LPN concentration. This was repeated for seven time points from 1/4 hour to 48 hours. The results are shown in table 2.6.5.5 in the report. The large number of animals required rules out a similar test on primates with a phsiology closer to humans.
It should be noted that in table 2.6.5.5B, when total LPN concentrations are measured, the liver followed by the spleen and the ovaries rank highest at 24.3, 23.4 and 12.3 (μg lipid equivalent / g [or mL]) after 48 hours respectively.
In the following 15:00 minute video Dr. Robert W. Malone, the inventor of mRNA vaccines, discusses the Pfizer data and explains the potential serious risks associated with the mRNA vaccines.
The speakers raise concerns over LPN (vaccine) concentration in the ovaries and also the bone marrow. They discuss short-term as well as long-term effects and concerns, underlining the need for phase 3 study followup for diseases and damage that does not clearly emerge for several years.
Ogata et al (2021)[36] found the S protein circulating in the blood stream of 13 subjects who received two doses of the Moderna vaccine. They note:
SARS-CoV-2 proteins were measured in longitudinal plasma samples collected from 13 participants who received two doses of [Moderna] mRNA-1273 vaccine.
Ogata et al (2021)[36]
In conclusion, in vivo or animal studies demonstrate the high PK mobility of the Pfizer vaccine, and presumably Moderna also, from the injection site throughout bodily tissues. Similar human results are beginning to appear for both the vaccine particles and S proteins but at this point we lack positive confirmation through controlled human autopsy studies.
An Overview of Associated Serious Adverse Effects
There are a number of serious adverse affects arising, actually or potentially, from the vaccines[32][48]. Dr. Roger Hodkinson identifies several in the following (15:31) minute video:
Some of the key points that Dr. Hodkinson makes are:
- With myocarditis, heart muscle cells are killed and these cells will not be regenerated. This can result in major cardiac problems later in life.
- A confidential Pfizer study that has been discussed above showed that in trials with rats, there was heavy accumulation of the S protein in the ovaries leading to concerns about female infertility.
- The ACE2 receptor which is the human cellular receptor that S primarily binds to is highly expressed in the placenta and testes, again leading to concerns about male fertility and female pregnancy including miscarriage.
- The S protein is responsible for a number of thrombotic events such as blood clots in the brain.
Following are sections addressing particular areas of concern.
Thrombosis (Blood Clotting)
Microvascular thrombotic disorders, very small blood clots on vascular system surfaces, have been reported from several sources[3][11][22][25][38]. This is leading to a conclusion stated by the Salk Institute that COVID-19 is primarily a vascular disease.
Dr. Malcolm Kendrick (2021)[25] gives a good discussion of vasculitis or inflammation of the blood vessels leading to blood clotting due to damage to the endothelial wall cells of the vascular system by the S protein.
Zhang et al (2020)[28] also note that the S protein binds to platelets resulting in clot formation and inflammation. They identify
platelet activation via binding of Spike to ACE2. SARS-CoV-2-induced platelet activation may participate in thrombus formation and inflammatory responses in COVID-19 patients
Zhang et al (2020)[28]
The key point is that the S protein is responsible for blood clot formation and inflammation throughout the body, including the brain.
Myocarditis and Pericarditi
A presentation from the CDC describes the condition and its treatment. It describes the incidence associated with vaccines, especially for males under 30, the group predominantly affected, as rare but notes in conclusion that “No long-term data available yet“[4].
In the US on June 25, the FDA added a warning to the Moderna and Pfizer vaccines:
Today, the FDA is announcing revisions to the patient and provider fact sheets for the Moderna and Pfizer-BioNTech COVID-19 vaccines regarding the suggested increased risks of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the tissue surrounding the heart) following vaccination.
FDA[2]
A military study by Montgomery et al (2021)[17] of a group of 23 mostly “healthy” males found:
[In] 23 male patients, including 22 previously healthy military members, myocarditis was identified within 4 days of receipt of a COVID-19 vaccine. For most patients (n = 20), the diagnosis was made after the second dose of mRNA COVID-19 vaccine
Montgomery et al (2021)[17]
Of note is that most of the cases arose after the second dose, consistent with what others have reported.
The media has been reporting regularly on young people, particularly males under 30, manifesting an unusual number of cases of myocarditis. The reader can find these by searching but we are generally not using sources other than scientific papers for this article.
Fertility and Pregnancy Concerns
Concerns by Drs Hodkinson, Malone, and Bridle expressed in the audio/video presentations have been discussed above in various sections of this article. There are many reports in the media which we don’t document, of miscarriages and menstrual cycle irregularities associated with the vaccines. These should raise serious concerns about the effects of both the vaccines and particularly the S protein on the female reproductive system and pregnancies.
In the meantime, we must depend on vaccine adverse effect reporting systems to look for abnormalreactions related to pregnancy[46].
Autoimmune Disease Concerns
Examples of autoimmune disease are narcolepsy, Guillain-Barré syndrome (GBS), multiple sclerosis, demyelinating neuropathies, systemic lupus erythematosus, and postural orthostatic tachycardia syndrome. Because we have no completed longitudinal studies, we have no idea if the vaccines will be responsible for a rise in such autoimmune diseases which can take up to years to manifest.
Researchers have raised concerns about autoimmune disease, particularly associated with the S protein[14][19]. Some sources reviewed in other parts of this article have also voiced such concerns.
Vojdani and Kharrazian (2021)[21] looked “for the potential cross-reactive interactions … between spike and nuclear proteins of SARS-CoV-2 and autoimmune target proteins“.
What this means is that they looked to see if antibodies created for the S protein by the immune system would also attack certain proteins of the human body. This is what creates an autoimmune disease. They found examples where such an autoimmune cross-reaction could occur within the cardiovascular, gastrointestinal, and nervous systems:
a sufficiently strong indication of cross-reaction between SARS-CoV-2 proteins and a variety of tissue antigens beyond just pulmonary tissue, which could lead to autoimmunity against connective tissue and the cardiovascular, gastrointestinal, and nervous systems.
Vojdani and Kharrazian (2021)[21]
They state their concern as:
The promotion and implementation of such an aggressive “immune passport” program worldwide in the absence of thorough and meticulous safety studies may exact a monumental cost on humanity in the form of another epidemic, this time a rising tide of increased autoimmune diseases …
Vojdani and Kharrazian (2021)[21]
Johnsom and Johnson have aded a notice alerting individuals to “rare cases of the neurological disorder, Guillain-Barré syndrome”[47].
In short, the vaccines are theoretically capable of creating autoimmune disorders. No studies have been made for such that we could find.
Neurodegenerative Implications
It is recognized that SARS-CoV-2 is associated with a number of central nervous system (CNS) and neurological pathologies (disorders), particularly of the brain’s vascular system[3][7][9][10][13][33][42]. Concerns have been raised that the spike protein may be directly responsible in part or in whole for these pathologies and the development of neurodegerative diseases such as Alzheimer’s Disease[10][12]. This section reviews the research and concerns related to the issue.
The Liver and the Blood-Brain Barrier
The brain is an organ of the body that has a unique protective measure. It is defensive mechanism known as the blood-brain barrier (BBB). The BBB should prevent both the vaccine components such as the nanolipid mRNA carriers and S proteins from entering the brain. Here is a somewhat technical explanation of the BBB.
A selectively permeable structural and functional barrier that exists between the capillaries and the brain; water, O2 and CO2 readily cross the BBB, glucose is slower, Na+, K+, Mg++, Cl–, HCO3– and HPO4– require 3-30-fold more time to equilibrate with the CSF than with other interstitial fluids; urea penetrates very slowly; catecholamines and bile salts essentially do not cross the BBB–kernicterus is due to accumulation of bile salts in the brains of neonates whose BBB is yet immature; integrity of the BBB is impaired in hepatic encephalopathy
Surce: The Free Dictionary and McGraw-Hill Concise Dictionary of Modern Medicine. © 2002 by The McGraw-Hill Companies, Inc.
It is the hepatic encephalopathy that is the key point. Impaired liver function leads to impaired BBB function and consequently opens up the possibility of a number of neurological disorders. Mild to moderate liver impairment has been noted in ~50% of COVID-19 cases. These studies also note the expression of the ACE2 receptor throughout the liver.
Licata et al. (2021)[43] echoed others when they noted:
Mild to moderate liver impairment during Covid-19 is common, especially in patients with pre-existing liver disease. Further studies should be performed in order to understand how pre-existing liver conditions may influence and worsen progression of liver disease in COVID-19 patients.
Licata et al. (2021)[43]
Jothimani et al. (2021)[44] placed the incidence of some impairment at up to 53%, a number that is consistent with other studies[45]. They also noted the prevalence of expression of the ACE2 receptor in the liver which we have shown to be the predominant source of damage to tissue due to S protein binding.
Buzhdygan et al (2020)[6] showed using in vitro models (lab-based experiments), that “the SARS-CoV-2 spike proteins trigger a pro-inflammatory response on brain endothelial cells that may contribute to an altered state of BBB function.” In other words, their models show that the spike protein may damage the BBB.
Animal studies have shown that the spike protein crosses the blood-brain barrier[5].
In summary, the S protein is responsible for damage to the blood-brain barrier that protects the brain from pathogenic materials that could cause brain disorders. The damage associated with S is both direct and indirect through damage to the liver. While the degree of potential liver involvement may seem surprising, given the concentration of vaccine LNPs in the livers of test animals it should not be.
Amyloid Binding Leading to Serious Neurodegenerative Diseases
Neurological effects in humans caused by the S protein are beginning to appear.
Idrees and Kumar (2021)[12] review a number of studies which they cite but we wont investigate since their work simply extends and confirms existing discoveries. They conclude:
In summary, the findings reported here support the hypothesis that the SARS-CoV-2 spike protein can interact with heparin binding amyloid forming proteins. Our results indicate stable binding of the S1 protein to these aggregation-prone proteins which might initiates aggregation of brain protein and accelerate neurodegeneration. These findings might explain the possible neurological distresses associated with COVID-19.
Idrees and Kumar (2021)[12]
They shows how the S protein can cause the aggregation of amyloid proteins in the brain causing Alzheimers and other neurodegenerative diseases. This supports the notion that the vaccines are seeding an epidemic of future debilitating neurological disorders.
Prion Disease Concerns
Prion diseases[24] are not viral in nature but purely protein-based. Certain proteins that naturally exist in the brain malfunction but have the property of being able to cause healthy proteins to malfunction in the same way. As the Government of Canada explains it:
The normal form of PrP (PrPc) is present in all healthy humans and animals. However, in prion disease somehow one or a few PrPc molecules are converted to an abnormal infectious form, or prion (proteinaceous infectious particle).
Government of Camada (2021)[23]
Idrees and Kumar (2021)[12 ] noted the possibility of the S protein binding to the prion binding protein in the brain invoking concerns of a form of prion disease. Others have raised similar concerns[18][32].
G. Tetz and V. Tetz (2020)[31] performed an in silico study (computer simulation) that “identified the presence of prion-like domains in the SARS-CoV-2 spike protein“. They noted:
a striking difference was observed in the distribution of prion-like domains in the spike protein, since SARS-CoV-2 was the only coronavirus with a prion-like domain found in the receptor-binding domain of the S1 region of the spike protein.
G. Tetz and V. Tetz (2020)[31]
Classen (2021b)[49] has raised similar concern based on the anlysis of British data.
In summary, there is enough evidence to raise serious concerns about the vaccines inducing long-term neurodegenerative and autoimmune disorders.
What Long COVID Might Tell US
Early on, a group of people emerged who experienced COVID-19 symptoms months after the infection had abated. These people became known as ‘long haulers’[40] and the condition known as ‘Long COVID’[41]. Although we lack complete longitudinal studies, we do have studies and reports on Long COVID.
It would be a reasonable line of research to examine their symptoms in the context of what is known about the potential long-term damage the S protein may cause. Some research into long-term effects is beginning to be conducted[39].
Discussion
Recent research is narrowing in on the spike protein as the major source of the damage done by COVID-19. It is clear that the S protein is a pathogenic toxin that can operate apart from either the vaccine LNP or the virus itself. It does so by binding to the ACE2 receptor expressed on epithelial and endothelial cells associated with all organs, systems and glands of the body. By binding, it down regulates or reduces the receptor function, disrupting the associated cellular processes leading to cell malfunction and cell death. It also creates microthrombi or blood clot and inflammation by activating a cytokine response from the immune system.
The vaccines deliver the mRNA or DNA to cells that then manufacture the toxic S protein. Evidence is that the vaccines do not stay at the injection site but travel throughout the body in the blood stream. By this, they create distributed local sites for S protein generation.
The Pfizer PK animal study provided evidence of the rapid distribution of the vaccine LNP active element throughout most bodily tissue. Normally such a discovery would inform the design of the phases of human trials. In particular, the human studies should have a focused on the concerns that have been raised in the literature and which have been identified in this report.
We have not seen any evidence in the public domain that the vaccine manufacturers were aware of vaccine mobility or S protein pathogenesis. That there was sufficient evidence to alert them is the case. We will not speculate why this information has been and continues to be suppressed. To this end, a note from Dr. Bridle indicates that he has suffered strong harassment.
It is disturbing that Pfizer in particular and likely the other manufacturers, along with health officials and authorities have suppressed critical medical information about the vaccines. This leads to speculation about possible motive and attendant conspiracy theories.
Recommendations
We have several recommendations:
- There should be a complete moratorium of all vaccination until proper phase 3 studies have been completed and the results thoroughly reviewed by the medical and scientific communities.
- A thorough risk analysis should be undertaken for every age cohort and cost/benefit studies should be conducted.
- The need for these vaccines at all should be reviewed considering that we have generic and cheap pharmaceuticals that when used in a proper protocol, provide excellent prophylaxis (prevention) and treatment. In particular, vitamin D3, hydroxychloroquine and Ivermectin have a large body of literature showing their efficacy.
- No person under 20 should receive the vaccines since there is no medical reason to do so. The risk outweighs potential benefits.
Summary
The vaccines in use are introducing a pathogenic toxin into the body whose immediate effect is to create permanent damage. In most cases it is minor. There are enough cases of seious and fatal adverse effects that individuals should be encouraged to do their own risk assessment. There are sufficiently serious potential long-term effects, however, yet to emerge which may see these vaccines as a medical disaster on a large scale.
In This Series
This article spawned a follow-up series of articles expanding the themes and updating existing material with new developments. Here is the series.
- Understanding the Danger of the SARS-CoV-2 Vaccines
- What Was the FDA Expecting from the Vaccines?
- Update #1: Delta Variant Lethality and COVID Vaccine Damage
- Update #2: COVID Vaccine Damage and Breakthrough Deaths
- Update #3: COVID Vaccine Damage – Graphene Oxide in the Pfizer Vaccine
- Update #4: COVID Vaccine Damage
- mRNA Vaccines: The Blood Clotting Killer
References
- SARS-CoV-2 mRNA Vaccine (BNT162, PF-07302048) 2.6.4 Summary of pharmacokinetic study. Pfizer. PDF.
- Coronavirus (COVID-19) Update: June 25, 2021. FDA. June 25, 2021.
- Chachques JC, Mazzini L, Mitrecic D, et al. Cardiovascular, hematological and neurosensory impact of COVID-19 and variants. Eur Rev Med Pharmacol Sci. 2021 Apr;25(8):3350-3364. doi: 10.26355/eurrev_202104_25747. PMID: 33928623.
- Oster M. Overview of Myocarditis and PericarditisACIP COVID-19 Vaccines Work Group. CDC. June 23, 2021. PDF.
- Rhea EM, Logsdon AF, Hansen KM, et al. The S1 protein of SARS-CoV-2 crosses the blood-brain barrier in mice. Nat Neurosci. 2021 Mar;24(3):368-378. doi: 10.1038/s41593-020-00771-8. Epub 2020 Dec 16. PMID: 33328624. PDF.
- Buzhdygan TP, DeOre BJ, Baldwin-Leclair A, et al. The SARS-CoV-2 spike protein alters barrier function in 2D static and 3D microfluidic in-vitro models of the human blood-brain barrier. Neurobiol Dis. 2020 Dec;146:105131. doi: 10.1016/j.nbd.2020.105131. Epub 2020 Oct 11. PMID: 33053430; PMCID: PMC7547916.
- Divani AA, Andalib S, Di Napoli M, et al. Coronavirus Disease 2019 and Stroke: Clinical Manifestations and Pathophysiological Insights. J Stroke Cerebrovasc Dis. 2020 Aug;29(8):104941. doi: 10.1016/j.jstrokecerebrovasdis.2020.104941. Epub 2020 May 12. PMID: 32689643; PMCID: PMC7214348.
- Farsalinos K, Eliopoulos E, Leonidas DD, et al. Cholinergic System and COVID-19: In Silico Identification of an Interaction between SARS-CoV-2 and Nicotinic Receptors with Potential Therapeutic Targeting Implications. Int J Mol Sci. 2020 Aug 13;21(16):5807. doi: 10.3390/ijms21165807. PMID: 32823591; PMCID: PMC7461543.
- Boroujeni ME, Simani L, Bluyssen HAR, et al. Inflammatory Response Leads to Neuronal Death in Human Post-Mortem Cerebral Cortex in Patients with COVID-19. ACS Chem Neurosci. 2021 Jun 16;12(12):2143-2150. doi: 10.1021/acschemneuro.1c00111. Epub 2021 Jun 8. PMID: 34100287; PMCID: PMC8204755.
- Zhou Y, Xu J, Hou Y, et al. Network medicine links SARS-CoV-2/COVID-19 infection to brain microvascular injury and neuroinflammation in dementia-like cognitive impairment. Alzheimers Res Ther. 2021 Jun 9;13(1):110. doi: 10.1186/s13195-021-00850-3. PMID: 34108016; PMCID: PMC8189279.
- Whelan P. Request for Comments related to consideration of vaccines against SARS-CoV-2. UCLA. December 08, 2020. PDF. Also US Government.
- Idrees D, Kumar V. SARS-CoV-2 spike protein interactions with amyloidogenic proteins: Potential clues to neurodegeneration. Biochem Biophys Res Commun. 2021 May 21;554:94-98. doi: 10.1016/j.bbrc.2021.03.100. Epub 2021 Mar 24. PMID: 33789211; PMCID: PMC7988450. Full text.
- Shimazawa R and Ikeda M. Potential adverse events in Japanese women who received tozinameran (BNT162b2, Pfizer-BioNTech). J of Pharm Policy and Pract 14, 46 (2021). https://doi.org/10.1186/s40545-021-00326-7. PDF.
- Bruno R, McCullough PA, Forcades i Vila T, et al. SARS-CoV-2 mass vaccination: Urgent questions on vaccine safety that demand answers from international health agencies, regulatory authorities, governments and vaccine developers. Preprint. PDF.
- Ljunggren D. Canada signs deals with Pfizer, Moderna for experimental COVID-19 vaccines. Reuters. August 05, 2020.
- Pfizer and BioNTech to supply Canada with their BNT162 mRNA-based vaccine candidate. Pfizer. August 05, 2020.
- Montgomery J, Ryan M, Engler R, et al. Myocarditis Following Immunization With mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol. Published online June 29, 2021. doi:10.1001/jamacardio.2021.2833.
- Classen JB. COVID-19 RNA Based Vaccines and the Risk of Prion Disease. Principia Scientific International. April 28, 2021.
- Talotta R. Do COVID-19 RNA-based vaccines put at risk of immune-mediated diseases? In reply to “potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases”. Clin Immunol. 2021 Mar;224:108665. doi: 10.1016/j.clim.2021.108665. Epub 2021 Jan 8. PMID: 33429060; PMCID: PMC7833091.
- Schoenmaker L, Witzigmann D, Kulkarni JA, Vet al. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int J Pharm. 2021 May 15;601:120586. doi: 10.1016/j.ijpharm.2021.120586. Epub 2021 Apr 9. PMID: 33839230; PMCID: PMC8032477.
- Vojdani A, Kharrazian D. Potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases. Clin Immunol. 2020 Aug;217:108480. doi: 10.1016/j.clim.2020.108480. Epub 2020 May 24. PMID: 32461193; PMCID: PMC7246018.
- Lei Y, Zhang J, Schiavon CR, et al. SARS-CoV-2 Spike Protein Impairs Endothelial Function via Downregulation of ACE 2. Circ Res. 2021 Apr 30;128(9):1323-1326. doi: 10.1161/CIRCRESAHA.121.318902. Epub 2021 Mar 31. PMID: 33784827; PMCID: PMC8091897.
- About Prion Diseases. Can. Gov. Accessed July 06, 2021.
- Prion Diseases. Johns Hopkins. Accessed July 06, 2021.
- Kendrick M. COVID19 – the spike protein and blood clotting. Blog. June 03,2021.
- Alexander PE, Dara P, Tenenbaum H. The COVID-19 spike protein may be a potentially unsafe toxic endothelial pathogen. June 01, 2021.
- Suzuki YJ, Gychka SG. SARS-CoV-2 Spike Protein Elicits Cell Signaling in Human Host Cells: Implications for Possible Consequences of COVID-19 Vaccines. Vaccines (Basel). 2021 Jan 11;9(1):36. doi: 10.3390/vaccines9010036. PMID: 33440640; PMCID: PMC7827936.
- Zhang S, Liu Y, Wang X, et al. SARS-CoV-2 binds platelet ACE2 to enhance thrombosis in COVID-19. J Hematol Oncol. 2020 Sep 4;13(1):120. doi: 10.1186/s13045-020-00954-7. PMID: 32887634; PMCID: PMC7471641.
- Avolio E, Gamez M, Gupta K. The SARS-CoV-2 spike protein disrupts the cooperative function of human cardiac pericytes – endothelial cells through CD147 receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. Madeddu bioRxiv 2020.12.21.423721; doi: https://doi.org/10.1101/2020.12.21.423721.
- Hansen T, Titze U, Kulamadayil-Heidenreich NSA,et al. First case of postmortem study in a patient vaccinated against SARS-CoV-2. Int J Infect Dis. 2021 Jun;107:172-175. doi: 10.1016/j.ijid.2021.04.053. Epub 2021 Apr 16. PMID: 33872783; PMCID: PMC8051011.
- Tetz G, Tetz V. SARS-CoV-2 Prion-Like Domains in Spike Proteins Enable Higher Affinity to ACE2. Preprints 2020, 2020030422 (doi: 10.20944/preprints202003.0422.v1). PDF.
- Seneff S, Nigh G. Worse Than the Disease? Reviewing Some Possible Unintended Consequences of the mRNA Vaccines Against COVID-19. International Journal of Vaccine Theory, Practice, and Research. 2(1), May 10, 2021.
- Philippens IHCHM, Böszörményi KP, Wubben JA, et al. SARS-CoV-2 causes brain inflammation and induces Lewy body formation in macaques. bioRxiv 2021.02.23.432474. May 05, 2021. doi: https://doi.org/10.1101/2021.02.23.432474
- Khan S, Shafiei MS, Longoria C, et al. SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway. bioRxiv [Preprint]. 2021 Mar 17:2021.03.16.435700. doi: 10.1101/2021.03.16.435700. PMID: 337588
- The novel coronavirus’ spike protein plays additional key role in illness. Salk News. April 30, 2021.
- Ogata AF, Cheng CA, Desjardins M, et al. Circulating SARS-CoV-2 Vaccine Antigen Detected in the Plasma of mRNA-1273 Vaccine Recipients. Clin Infect Dis. 2021 May 20:ciab465. doi: 10.1093/cid/ciab465. Epub ahead of print. PMID: 34015087; PMCID: PMC8241425.
- Samavati L, Uhal BD. ACE2, Much More Than Just a Receptor for SARS-COV-2. Front Cell Infect Microbiol. 2020 Jun 5;10:317. doi: 10.3389/fcimb.2020.00317. PMID: 32582574; PMCID: PMC7294848.
- Phend C. COVID Heart Autopsies Point More to Clot Damage Than Myocarditis. MedPage Today October 15, 2020.
- Ries J. What We Know About COVID-19 and Long-Term Heart Damage. Healthline. October 19, 2020.
- Powell A. A pandemic that endures for COVID long-haulers. Harvard Gazette. April 13, 2021.
- Masterson V. What is long COVID? WEF. July 01, 2021.
- Najjar S, Najjar A, Chong DJ, et al. Central nervous system complications associated with SARS-CoV-2 infection: integrative concepts of pathophysiology and case reports. J Neuroinflammation. 2020 Aug 6;17(1):231. doi: 10.1186/s12974-020-01896-0. PMID: 32758257; PMCID: PMC7406702.
- Licata A, Minissale MG, Distefano M, et al. Liver injury, SARS-COV-2 infection and COVID-19: What physicians should really know? GastroHep. 2021 May;3(3):121-130. doi: 10.1002/ygh2.455. Epub 2021 May 3. PMID: 34149320; PMCID: PMC8207035.
- Jothimani D, Venugopal R, Abedin MF, et al. COVID-19 and the liver. J Hepatol. 2020 Nov;73(5):1231-1240. doi: 10.1016/j.jhep.2020.06.006. Epub 2020 Jun 15. PMID: 32553666; PMCID: PMC7295524.
- Elhence A, Vaishnav M, Biswas S, et al. Coronavirus Disease-2019 (COVID-19) and the Liver. J Clin Transl Hepatol. 2021 Apr 28; 9(2): 247–255. Published online 2021 Mar 22. doi: 10.14218/JCTH.2021.00006.
- Lawrie T. RE: Urgent preliminary report of Yellow Card data up to 26th May 2021. EbMC Squared CiC. June 09, 2021. PDF.
- Johnson & Johnson Statement on COVID-19 Vaccine (7/12). J&J. July 12, 2021.
- An Evidence Review from the Penn Medicine Center for Evidence-based Practice. CEP. December 2020. PDF.
- Classen JB (2021b). COVID-19 Vaccine Associated Parkinson’s Disease, A Prion Disease Signal in the UK Yellow Card Adverse Event Database. J Med – Clin Res & Rev. 2021; 5(7): 1-6. July 18, 2021. PDF.
1 Comment
save refuges · June 21, 2022 at 12:32 am
Hi there mates, its impressive article regarding educationand entirely defined, keep it up all the time.
save refuges