Last updated by The POOG on May 05, 2021.
There has been a world-wide rush to get the first vaccine to market. It’s similar to a gold rush, both in the frantic haste and in the enormous wealth that is at stake to successful participants. However, serious questions surround these vaccines and the development process. We explore them in this article. At the end is a video by James Corbett on mRNA vaccines and where the technology is headed. If nothing else, watch this video.
Phases of Vaccine Development and Testing
The first phase is vaccine design and formulation. Testing stages are in vitrio (cell culture), in vivo (animal studies), and three stages of human testing. ZeroHedge gives a good overview of the phases and normal duration of standard vaccine development versus COVID-19 vaccines[18]. Due to mRNA technology, the design stage of mRNA vaccines may be considerately shorter than standard antigen or viral vector vaccines.
mRNA Vaccine Technology
A virus is a complex of organic molecules, one of which is either a strand of DNA or RNA. These molecular strands are related and contain the genetic code necessary to reproduce the virus. The family of corona viruses are RNA viruses meaning that their genetic code is in the form of a strand of RNA called messenger RNA (mRNA).
The virus reproduces by attaching to a human body cell, and inserting its mRNA into the cell where the cellular reproduction engine is hijacked to reproduce copies of the virus encoded by the viral mRNA. The cell recognizing that it has been infected, commits suicide releasing the new virus copies into the surrounding medium where they repeat the process.
The Simple Description
The mRNA vaccines operate like a virus. They encapsulate a fragment of the virus mRNA that encodes the virus spike protein, in a lipid (fatty) nanoparticle (LNP). The LNP acts like the virus by binding to a cell and insert the mRNA fragment into the cell. The cellular reproduction engine creates copies of the spike protein encoded by this mRNA fragment. These are recognized by the cell as foreign and are expelled by moving them to the cellular surface where the immune system can find and identify them. It can then create antibodies to inactivate any virus encountered outside of cells.
The following video by Shane Crotty gives an excellent discussion of the technology and how it functions.
Here is a diagram showing the process described above.
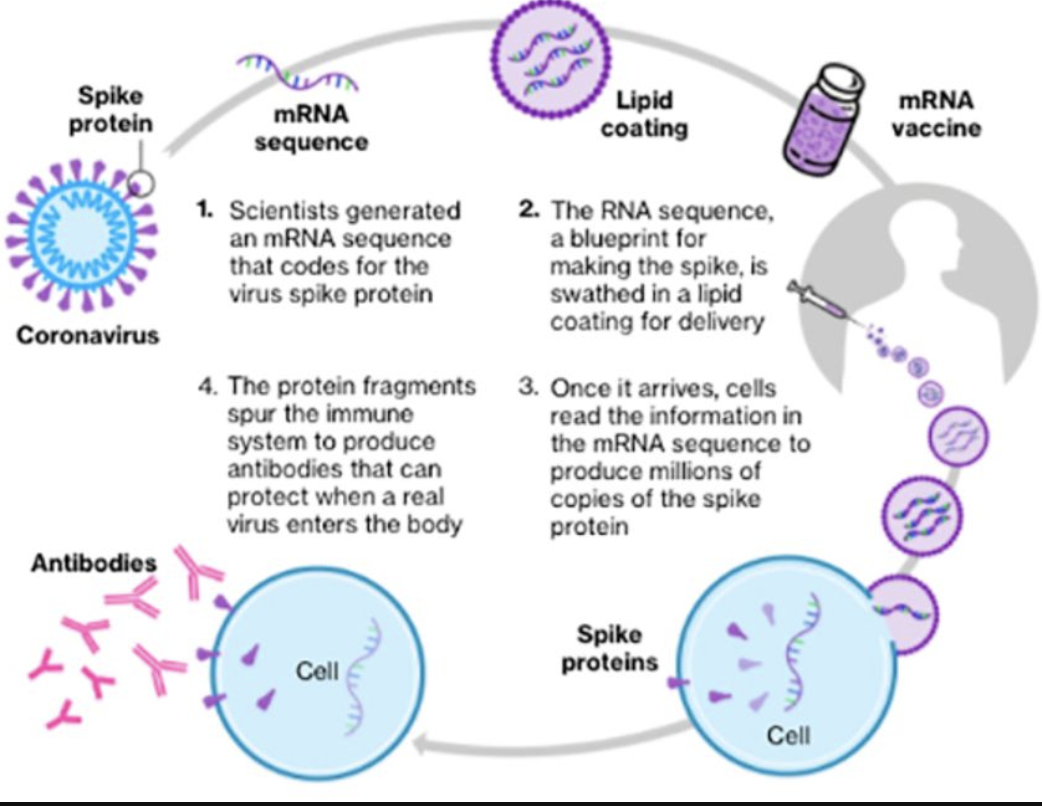
Highly Technical Descriptive Material
This material is collected for in-depth analysis by people with a background which includes such fields as biochemistry and molecular biology.
The Cottrell Series
Dr. Paul Cottrell has announced a series of videos analyzing the mRNA vaccine. It is in the form of a set of videos which we will provide links to as they become available.
- The first video explains the details of a paper that describes the biochemistry of the lipid nano-particles (LNP) that the mRNA is embedded in as a means of introducing it into the cell: https://www.youtube.com/watch?v=-yJVRyLivjw.
- The second video describes how Moderna engineered the extraction of viral RNA and modified it for insertion into an LNP so that the vaccine itself could be injected without triggering the immune system. https://www.youtube.com/watch?v=YjI0pbJiHh4.
Read for Risk Analysis and Legal Proceedings
Sadaf Gilani[30] has presented an overview of the technical issues and concerns with the mRNA ‘vaccines’ and of the way they have been rolled out. Individuals given either vaccines are unlikely to be informed of the full risks involved and if they are, are unlikely to fully comprehend them. If you are involved in a legal proceeding over the vaccine in some capacity, this should be required reading.
Candidate Vaccines
These are vaccines currently in development or in early roll-out. Relevant technical data and studies are captured. The intent is to provide you with enough facts to inform your decision around taking a vaccine. Here is a list of current vaccine projects as of December 2020:
AstraZeneca ChAdOx1 nCoV-19 (AZD1222) Vaccine
Oxford University vaccine candidate AZD1222. Not yet researched.
News Updates
Discussion of the Vaccine
Efficacy (Effectiveness)
Voysey et al (2020)[16] reported interim results from the first clinical trials of ChAdOx1 nCoV-19 and found the the following results:
In participants who received two standard doses, vaccine efficacy was 62·1% (95% CI 41·0–75·7; 27 [0·6%] of 4440 in the ChAdOx1 nCoV-19 group vs71 [1·6%] of 4455 in the control group) and in participants who received a low dose followed by a standard dose, efficacy was 90·0% (67·4–97·0; three [0·2%] of 1367 vs 30 [2·2%] of 1374; pinteraction=0·010).
Voysey et al (2020)[16]
This is a curious result because better results arose from the group that received a low dose followed by a standard dose, than from the group that received two standard doses.
Updated phase III preliminary results lowered the efficacy to 76%[32].
Contraindications (Not Recommended For)
Side Effects
I have seen no formal reports to date but isolated reports are emerging[31].
Administration Protocols
Johnson & Johnson-Jannsen Ad26.COV2.S Vaccine
Ad26.COV2.S is a single dose vaccine stored at room temperature.
Efficacy (Effectiveness)
A company news release[29] gives preliminary trial results:
Vaccine Candidate 72% Effective in the US and 66% Effective Overall at Preventing Moderate to Severe COVID-19, 28 Days after Vaccination.
85% Effective Overall in Preventing Severe Disease and Demonstrated Complete Protection Against COVID-19 related Hospitalization and Death as of Day 28
J&J news release[29].
Moderna mRNA-1273 Vaccine
The Moderna COVID-19 vaccine is described by the FDA as
a white to off-white, sterile, preservative-free frozen suspension for intramuscular injection. The vaccine contains a synthetic messenger ribonucleic acid (mRNA) encoding the pre-fusion stabilized spike glycoprotein (S) of SARS-CoV-2 virus. The vaccine also contains the following ingredients: lipids (SM-1The candidate vaccine mRNA-1273 is an mRNA vaccine that encodes the stabilized prefusion SARS-CoV-2 spike protein.
FDA[10]
The initial trial[27] conducted was a
phase 1, dose-escalation, open-label trial including 45 healthy adults, 18 to 55 years of age, who received two vaccinations, 28 days apart, with mRNA-1273 in a dose of 25 μg, 100 μg, or 250 μg. There were 15 participants in each dose group.
Jackson et al. (2020)[23]
Phase 2 trials began in May using 25-μg and 100-μg doses. The latter is the dose used in phase 3 trials and public application. A 250-μg dose in phase 1 trials created severe adverse reactions and was not used in phase 2[23][24].
The FDA Briefing Document gives a good overview of the vaccine and phase 3 trials[10].
News Updates
Discussion of the Vaccine
Excipients (additives) to the vaccine include[5]:
- lipid: (SM-102, 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000 [PEG2000-DMG]
- lipid: cholesterol
- lipid: 1,2-distearoyl-sn-glycero-3-phosphocholine [DSPC])
- tromethamine,
- tromethamine hydrochloride,
- acetic acid,
- sodium acetate, and
- sucrose.
Efficacy (Effectiveness)
Early analysis[10] shows a 94% – 95% efficacy. As one paper notes[25]:
Primary efficacy analysis of the Phase 3 COVE study of mRNA-1273 involving 30,000 participants included 196 cases of COVID-19, of which 30 cases were severe Vaccine efficacy against COVID-19 was 94.1%.
Source: Webull[25]
Immunity
Widge et al (2020)[26] reported from the phase 3 trials:
At the 100-μg dose, mRNA-1273 produced high levels of binding and neutralizing antibodies that declined slightly over time, as expected, but they remained elevated in all participants 3 months after the booster vaccination.
Widge et al (2020)[26]
Contraindications (Not Recommended For)
information not yet found
Side Effects
Side effects for the phase 1 trials included below in Table 1 are not quantified as in the case of the Pfizer vaccine.

It should be noted that all participants experienced mild to moderate pain and other “local” and “systemic” symptoms. There were more symptoms after the second dose and of greater severity. When you think of the standard flu vaccine, this vaccine with sme effects affecting 100% of recipients is not as benign.
Anecdotal and News Media Reports
Until scientific studies appear on vaccine side effects we have to rely on this type of reporting. Examples of severe effects appearing in the media are available[21][22].
Administration Protocols
This vaccine has very particular temperature requirements[10].
The Moderna COVID-19 Vaccine is provided as a frozen suspension [stored between -25º to -15ºC (-13º to 5ºF)] multi-dose vial containing 10 doses. The vaccine must be thawed prior to administration. After thawing, a maximum of 10 doses (0.5 mL each) can be withdrawn from each vial. Vials can be stored refrigerated between 2° to 8°C (36° to 46°F) for up to 30 days prior to first use. Unopened vials may be stored between 8° to 25°C (46° to 77°F) for up to 12 hours. After the first dose has been withdrawn, the vial should be held between 2° to 25°C (36° to 77°F) and discarded after 6 hours.
FDA[10].
The Moderna COVID-19 vaccine, mRNA-1273 (100 μg) is administered intramuscularly as a series of two doses (0.5 mL each), given 28 days apart.
Novavax, Inc.
Not yet researched.
Pfizer-BioNTech BNT162b2 Vaccine
The FDA Briefing Document[5] gives a detailed overview of the vaccine and various phases of trials.
News Updates
These are short references mostly of a news nature to track developments.
20201208. FDA Says 2 Participants In Pfizer COVID Vaccine Trial Have Died[15]. I include this articles as an instructional item. First, it has no information about general health, cause of death, or any association with the vaccines. These are two people out of a test group of 30,000. Let’s say they had the vaccine a week before. How many people die each week in a city of 30,000? Read the obits. The point is there will be deaths of recipients unrelated to the vaccine.
Discussion of the Vaccine
BNT162b2 is an mRNA vaccine with the RNA embedded in lipid nanoparticles. It requires a 2-dose regimen administered 21 days apart. The UK government gave this description:
COVID-19 mRNA Vaccine BNT162b2 is highly purified single-stranded, 5’-capped messenger RNA (mRNA) produced by cell-free in vitro transcription from the corresponding DNA templates, encoding the viral spike (S) protein of SARS-CoV-2.
Source: UK government[8]
Excipients (additives) to the vaccine include[8]:
- lipid: ALC-0315 = (4-hydroxybutyl) azanediyl)bis (hexane-6,1-diyl)bis(2-hexyldecanoate)
- lipid: ALC-0159 = 2-[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide
- lipid: 1,2-Distearoyl-sn-glycero-3-phosphocholine
- lipid: cholesterol
- potassium chloride
- potassium dihydrogen phosphate
- sodium chloride
- disodium hydrogen phosphate dihydrate
- sucrose
- water for injections
Efficacy (Effectiveness)
The initial report by Pfizer gave an efficacy of 94.6% based on a study size of 40,000 participants. The sample size of participants reviewed for this estimate is small. An FDA Briefing Document[5] confirms this figure.
It should be noted that the study will continue for 2 years[8].
Contraindications (Not Recommended For)
- Individuals receiving anticoagulant therapy or those with a bleeding disorder that would contraindicate intramuscular injection[8].
- Immunocompromised persons, including individuals receiving immunosuppressant therapy, may have a diminished immune response to the vaccine[8].
- Any person with a history of immediate-onset anaphylaxis to a vaccine, medicine or food should not receive the COVID-19 mRNA Vaccine BNT162b2[8][11].
- Hypersensitivity to the active substance or to any of the excipients listed above.
- Individuals with a history of severe allergic reactions but not to vaccines should consult there doctor before proceeding[12].
Side Effects
In listing side effects, the following definitions apply[8]:
- very common (≥ 1/10)
- common (≥ 1/100 to < 1/10)
- uncommon (≥ 1/1,000 to < 1/100)
- rare (≥ 1/10,000 to < 1/1,000)
- very rare (< 1/10,000)
- not known (cannot be estimated from available data)
For the Pfizer BNT162b2 vaccine, effects are:
Very common
- Arthralgia; myalgia[8]
- Headache[8]
- Injection-site pain; fatigue; chills; pyrexia[8]
Common
- Redness at injection site; injection site swelling[8]
- Nausea[8]
Uncommon
- Lymphadenopathy[8]
- Malaise
Rare
- Anaphylactic reaction: severe anaphylactic reaction has been reported in the US[7][11] and the UK[9]. “Persons with anaphylaxis following COVID-19 vaccination should not receive additional doses of COVID-19 vaccine“[11]][28].
Areas of Concerns (usually no studies performed)
- Effect on female/male fertility (The UK guidelines[8] notes “Animal studies into potential toxicity to reproduction and development have not been completed“.)
- Effect on pregnant women (recommendation advises against getting pregnant within 2 months of the last dose)
- Effect on breast feeding
- Interaction with other vaccines or drugs
- Pathenogenic priming. This is potentially serious enough to warrant a separate section below.
Anecdotal and News Media Reports
Administration Protocols
The proposed dosing regimen is 2 doses, 30 μg each, administered 21 days apart[5].
Persons administering the vaccine must be trained and prepared to deal with an Anaphylactic reaction[9].
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
Pathogenic Priming
Because there is a high homology (similarity) between the corona virus spike proteins and proteins native to the human body, there is a risk that developing a vaccine against SARS-CoV-2 by using parts of the spike protein that are homologous to human proteins. Such may prime the immune system to create an autoimmune response[13][14] when the immune system at a later date is presented with the virus. This could establish an autoimmune disease in the recipient. As Lyons-Weiler (2020)[13] notes:
Failure of SARS and MERS vaccines in animal trials involved pathogenesis consistent with an immunological priming that could involve autoimmunity in lung tissues due to previous exposure to the SARS and MERS spike protein. Exposure pathogenesis to SARS-CoV-2 in COVID-19 likely will lead to similar outcomes.
Lyons-Weiler (2020)[13]
A PubMed search didn’t reveal any other relevant papers. However the possibility of induced prion disease is a concern: STUDY: COVID-19 RNA Based Vaccines and the Risk of Prion Disease.
University of Queensland-CSL UQ-CSL V451 Vaccine
Phase one clinical trials of UQ-CSL V451 returned false positive test results for HIV. The participants who tested positive did not have an HIV infection but carried HIV homologous fragments from the vaccine that could confuse testing of future recipients.
The project has been terminated[6].
Anecdotal Reports of Side Effects
Until scientific studies appear on vaccine side effects we have to rely on this type of reporting. A Pfizer[17] news release gave side effects as:
To date, the Data Monitoring Committee for the study has not reported any serious safety concerns related to the vaccine. A review of unblinded reactogenicity data from the final analysis which consisted of a randomized subset of at least 8,000 participants 18 years and older in the phase 2/3 study demonstrates that the vaccine was well tolerated, with most solicited adverse events resolving shortly after vaccination. The only Grade 3 (severe) solicited adverse events greater than or equal to 2% in frequency after the first or second dose was fatigue at 3.8% and headache at 2.0% following dose 2. Consistent with earlier shared results, older adults tended to report fewer and milder solicited adverse events following vaccination.
Pfizer[17]
Examples of moderate and severe effects appearing in the media are available[19][20]. A summary of effects of the Pfizer vaccine is given as[8][5]:
Unfortunately, none of the terms are defined or quantified so the results cannot be matched against the side effects described above. Note that children were not tested.
The most frequent adverse reactions in participants 16 years of age and older were pain at the injection site (> 80%), fatigue (> 60%), headache (> 50%), myalgia (> 30%), chills (> 30%), arthralgia (> 20%) and pyrexia (> 10%) and were usually mild or moderate in intensity and resolved within a few days after vaccination.
Source: UK government[8]
When you think of the standard flu vaccine this vaccine with effects affecting 80% of recipients is very high. The safety profile of BNT162b2 was generally similar across all categories of participants in the study[5]
Here’s one person experience of a severe side effect to Moderna.
The Dystopian Future of Vaccines and mRNA Technology
Vaccines for SARS-CoV-2 have become a geopolitical issue for some individuals and organization. They are using vaccines as one part of a strategy to extend control over their populations. James Corbett has done an excellent video covering the history of vaccines and the associated systems that are being developed along side them for population tracking and control.
Reprogramming Your DNA
Corbett had touched on this but I had written this earlier so am keeping it. Recent research has found a way to use messenger RNA (mRNA), a critical part of your cellular biology, to reprogram your DNA in its role of cellular reproduction of your life-form. Raul Diego[1], describes it as a
technology, that allows for biological processes to be controlled remotely and opens the door to the potential manipulation of our biological responses and, ultimately, our entire existence, is called Profusa Inc and its operations are funded with millions from NIH and DARPA [Defence Advanced Research Projects Agency].
Source: Raul Diego[1]
The technology was developed as an implantable health monitoring system[4] in the form of an implantable chip or injectable hydrogel. Such may be reprogrammed remotely using technologies such as 5G networks[2]. This technology may be used as the delivery system for mRNA vaccine[3] and other future vaccines.
Critical Concerns
There are many healthcare professionals, both individually and in groups that have expressed a range of concerns about these vaccines, both in their nature and administration. We capture some of the links here[33].
References
- Raul Diego. A DARPA-Funded Implantable Biochip to Detect COVID-19 Could Hit Markets by 2021. MPN News, September 17th, 2020.
- Patrick Tucker. A Military-Funded Biosensor Could Be the Future of Pandemic Detection. Defense One, March 3, 2020.
- Makia Freeman. Hydrogel Biosensor: Implantable Nanotech to be Used in COVID Vaccines? The Mad Truther, September 4, 2020.
- Col. Matt Hepburn. DARPA SBIR: Profusa Implantable Biosensors – COL Matt Hepburn. YouTube, 11 Oct 2018.
- Vaccines and Related Biological Products Advisory Committee Meeting. US FDA, December 10, 2020.
- Melissa Davey. Australia terminates University of Queensland vaccine deal with CSL after false positives for HIV. The Guardian, December 10, 2020.
- Pfizer to assess report about ‘potential serious allergic reaction’ to Covid-19 vaccine after Alaska health worker is hospitalized. RT, December 16, 2020.
- Information for Healthcare Professionals on Pfizer/BioNTech COVID-19 vaccine. UK Government, Updated 16 December 2020.
- Confirmation of guidance to vaccination centres on managing allergic reactions following COVID-19 vaccination with the Pfizer/BioNTech vaccine. UK Government, Updated 09December, 2020.
- Moderna COVID-19 Vaccine. US FDA, December 17, 2020.
- Clark T. Anaphylaxis Following m-RNA COVID-19 Vaccine Receipt. CDC. December 19, 2020.
- COVID-19 Vaccines and Severe Allergic Reactions. CDC. Updated Dec. 19, 2020.
- Lyons-Weiler J. Pathogenic priming likely contributes to serious and critical illness and mortality in COVID-19 via autoimmunity. J Transl Autoimmun. 2020 Apr 9;3:100051. doi: 10.1016/j.jtauto.2020.100051. eCollection 2020.
- Lyons-Weiler J and Kennedy RFjr. Pfizer COVID Vaccine Trial Shows Alarming Evidence of Pathogenic Priming in Older Adults. the Defender. December 10, 2020.
- Editors. FDA Says 2 Participants In Pfizer COVID Vaccine Trial Have Died. ZeroHedge. Tuesday, Dec 08, 2020.
- Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. December 08, 2020. DOI:https://doi.org/10.1016/S0140-6736(20)32661-1.
- Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints. Pfizer. November 18, 2020.
- Editors. Here Is Everything We Know About A COVID-19 Vaccine. ZeroHedge. September 4, 2020.
- TVR Staff. Pfizer/BioNTech’s COVID-19 Vaccine Causes Adverse Reactions in Over Half of Clinical Trial Volunteers. The Vaccine Reaction. July 18, 2020.
- Editors. COVID-19 ‘Phase 3’ Vaccine Trial Participants Report Day-Long Migraines, Fever. ZeroHedge. October 01, 2020.
- Cáceres M. Volunteer Describes His Serious Reaction in Moderna’s mRNA COVID-19 Vaccine Trial. The Vaccine Reaction. May 30, 2020.
- TVR Staff. Moderna’s COVID-19 Vaccine Provokes ‘COVID-Like Symptoms’ in Phase 3 Trial. The Vaccine Reaction. October 19, 2020.
- Jackson LA, Anderson EJ, Rouphael NG, et al. An mRNA Vaccine against SARS-CoV-2 — Preliminary Report. N Engl J Med; 383:1920-1931. November 12, 2020. DOI: 10.1056/NEJMoa2022483.
- Moderna Announces First Participants in Each Age Cohort Dosed in Phase 2 Study of mRNA Vaccine (mRNA-1273) Against Novel Coronavirus. Moderna. May 29, 2020.
- Moderna Announces Primary Efficacy Analysis In Phase 3 COVE Study For Its COVID-19 Vaccine Candidate; Vaccine Efficacy Against COVID-19 Was 94.1%, Vaccine Efficacy Against Severe COVID-19 Was 100%. Webull. November 30, 2020.
- Widge A, Rouphael NG, Jackson LA, et al. Durability of Responses after SARS-CoV-2 mRNA-1273 Vaccination. NEJM. December 3, 2020. DOI: 10.1056/NEJMc2032195.
- Safety and Immunogenicity Study of 2019-nCoV Vaccine (mRNA-1273) for Prophylaxis of SARS-CoV-2 Infection (COVID-19). NIH. Update: December 21, 2020.
- Staff. Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine — United States, December 14–23, 2020. CDC. January 6, 2021.
- News release. Johnson & Johnson Announces Single-Shot Janssen COVID-19 Vaccine Candidate Met Primary Endpoints in Interim Analysis of its Phase 3 ENSEMBLE Trial. J&J. January 29, 2021.
- Gilani S. A number of internet tools. Off Guardian. February 22, 2021.
- Allen A and Szabo L. NIH Concerned About COVID Vaccine Side Effects. WebMD. September 14, 2020.
- AZD1222 US Phase III primary analysis confirms safety and efficacy. AstraZeneca. 25 March 2021.
- Doctors for Covid Ethics. COVID Vaccines: Necessity, Efficacy and Safety. OffGuardian.May 5, 2021 .